Science
Related: About this forumCan We Recover Carbon Dioxide From the Atmosphere Using Sargassum Seaweed?
I will discuss two scientific papers in this post, one somewhat more extensively than the other. They are:
Biofuel production through micro- and macroalgae pyrolysis – A review of pyrolysis methods and process parameters. (Sara Pourkarimia, Ahmad Hallajisanib, Asghar Alizadehdakhela, and Amideddin Nouralishahib, corrected proof in press, Journal of Analytical and Applied Pyrolysis, 2019)
...and...
Microwave-assisted pyrolysis and analytical fast pyrolysis of macroalgae: product analysis and effect of heating mechanism (Ribhu Gautam, S. Shyam, B. Rajasekhar Reddy, Kasivelu Govindarajub and R. Vinu,* Sustainable Energy and Fuels, Advanced Release, 2019)
All efforts to address climate change have failed, and done so dramatically. It is somewhat ironic that as the crisis is now becoming so obvious that it cannot really stand up to repeated denial, that there has been a world wide renaissance of quasi-fascist or actually fascist ignoramuses advanced into political power in major and not so major countries, including not just the United States, but also the Philippines, Brazil, Hungary, Turkey and most recently Great Britain.
It doesn't bode well.
We, of course, on the left are not innocent in this outcome. We have, in general, although I have personally and certainly been disagreeing vehemently for quite some time, participated in handing out ignorance on our end of the political spectrum, this being the absurd and frankly stupid arguments against nuclear power claiming it's "dangerous" when the death toll associated with nuclear energy is absurdly minuscule when compared with the enormous death toll associated with the dangerous fossil fuel waste in the air, and is about to rise because heat deaths will begin to accelerate and begin to approach the death toll from other forms of dangerous fossil fuel waste and dangerous biomass. These far more deadly forms of energy kill by the formation of particulates, (especially PM10), carcinogens (aromatic PAH's, benzofurans, benzodioxins), carbon monoxide, sulfur oxides, nitrogen oxides (some of which are ozone depletion agents), and let's not forget our friends, the heavy metals, which are an inevitable consequence of fossil fuel combustion. (The world exposure to mercury from vaccines, the target of many ignoramuses, is dwarfed by orders of magnitude by exposure from coal combustion.) These latter types of deaths - excluding heat deaths - are estimated to kill about 7 million people per year, with slightly less than half of them attributed to the combustion of biomass and slightly more than half attributed to dangerous fossil fuels.
The absurdity of the argument that "nuclear power is dangerous" is so stupid by comparison to the obvious health effects of fossil fuels and the obvious effects of climate change from the dangerous fossil fuel waste carbon dioxide, that is should be obvious by inspection but as is the case of other forms of popular ignorance, Trumpism, Brexitism, blah, blah. If I didn't have an ignore button here, I'm sure I'd have to read tripe from these kinds of people, from the kinds of asses who write stuff - this actually happened - about the collapse of a tunnel at the Hanford weapons plant being a tragedy, while the heat deaths across Europe is regrettable but acceptable.
These kinds of people actually exist. Incredible, but they do.
Anyway, despite my hostility to so called "renewable energy" - such hostility arising from the fact that these fantasies, despite absorbing money on a multi-trillion dollar scale they have not done a damned thing to address climate change, are not doing any to address climate change and won't do anything to address climate change, I am about to write about biofuels (and while not explicitly stated) biomaterials.
Right now, not in some far off future getting the attention of the likes of say, Bill McKibben, anti-nuke "concerned" about climate change, or other weak minded people like say, Joe Romm, another shit for brains anti-nuke "concerned" about climate change, a massive mat of the macroalgae known as Sargassum, which used to be fairly limited to the Sargasso Sea in the Atlantic, now stretches across the entire ocean. While such outbreaks have a huge potential to kill the seas by the same mechanism that has killed the ecology of the Mississippi River Delta, by depleting oxygen when the algae dies and is decomposed by bacteria, it occurs to me that these macroalgae contain a lot of carbon, from the world's largest, by far, dump for the dangerous fossil fuel waste carbon dioxide, the ocean.
So I decided to look for recent articles - limiting the search to 2019 - to studies of the pyrolysis of sargassum, picking, at random, the two aforementioned papers from the collection I was able to download in about 10 minutes time.
"Pyrolysis" is the practice of heating mixtures of carbon compounds - biomass is an obvious case of such a mixture, although there are others, waste plastics for instance - in the absence of air, or at least oxygen, a temperatures at which they decompose to useful chemicals. This is hardly a new process. A very long time ago, when I was a child, one could buy in hardware stores cans of "wood alcohol" which was essentially methanol, the C1 alcohol, H3COH. Methanol used to be made by the pyrolysis of wood, although today it is largely made from carbon monoxide obtained from dangerous natural gas.
Although one can obtain biofuels from a number of processes, the silliest being one of the most widely used, fermentation, a water and energy intensive process, other processes exist and in my view pyrolysis and the related practice of supercritical water or supercritical carbon dioxide oxidation are, unlike every other so called "renewable energy" scheme, possibly sustainable (under the right conditions) and in fact are the low hanging fruit for the removal of carbon dioxide from the atmosphere. This removal of carbon dioxide, an incredible engineering challenge, may prove a matter of survival for future generations, about whom we clearly couldn't care less while we blithely carry on mindlessly about how they'll all drive Tesla cars some day.
The processes for obtaining biofuels (and more importantly biomaterials although they are scarcely mentioned in these papers) are shown in this graphic from the first paper cited:

The caption:
Here by the way, is an excerpt from the authors' introductory text in this review paper:
Third generation biofuels mainly derived from algal biomass are the most promising alternative resource without the drawbacks of first- and second-generation biofuels [14]. Algae are simple aquatic organisms living in fresh or saline water and are able to convert sunlight, water, and carbon dioxide into algal biomass photosynthetically. Due to many advantages, such a fast growth rate, CO2 fixation ability, possibility to grow in non-arable lands or in saline and wastewaters and no competition with food/feed crops, algal biomasses appears to be ideal third generation biofuel feedstocks [15]. However, large-scale production of algal biomass and cost efficiency generation of final bio-fuel products are the main problems associated with these biomass resources. It has been estimated that the average cost of biodiesel generated from microalgae is seven times higher than the current price of diesel [13]. Conversion of algal biomass into valuable energy products can be performed mainly by thermochemical, biochemical and chemical pathways [16,17]. Thermochemical conversion techniques use heat and chemical catalysts to produce the energy in three forms of bio-oil, biochar, and gaseous products while chemical/biochemical processes form mainly bio-diesel, ethanol and hydrogen [4,18]. Among thermochemical conversion processes, pyrolysis is a popular approach wherein the thermal degradation of the biomass components is performed in an inert atmosphere [19]. Pyrolyzed products in the form of bio-oil, biochar and gas can be used as fuel for energy generation. Besides fuel resource, biochar can be effectively used in agriculture as fertilizer to upgrade yield and plant nutrition [20–22], and in other fields to remove heavy metals [23–27], dyes [28] and other organic, inorganic and microbial pollutants [29,30].
I will focus in this discussion of sargassum for two reasons, one being that it is here in now disturbing quantities and the other being that it, like coral, tends to concentrate uranium from seawater, (along with other metals), the quantities of uranium being present in seawater is on the order of five billion tons. This means that the sargassum - given its huge mass - is a potential low grade (but workable) uranium ore when the carbon is removed from it. Thus potentially it contains the enormously high energy density fuel that might be used to pyrolyze it, much of the cost of pyrolysis being driven by the heat to affect it. Also sargassum is a macroalgae, meaning that it is easy to remove from seawater with simple nets; it also is likely to capture mechanically or internally, large amounts of the plastic waste that now contaminates the ocean.
If you don't know what sargassum is, here's a picture of it:

If you've been to the ocean, you've probably seen it lots of times.
The elemental composition by weight of sargassum is given in this first paper, 25.9% carbon, 5.57% hydrogen, 24.18% oxygen, 3.58% nitrogen, 1.22% sulfur. Among species of algae, it has the lowest high heating value, 8.68 MJ/kg, but this should not really matter unless the goal were direct combustion, a dangerous process I would oppose.
In terms of the biochemistry, sargassum is said to be (in this paper) about 10.25% protein, 0.74 lipids, and 41.81% carbohydrates. Possibly the balance, not specified, is represented by lignins. The low lipid content accounts for the low heating value, which means this particular species would not be good for producing biodiesel, although there is no particular advantage of biodiesel compared to other potential fuels, particularly those derived from carbohydrates, notably the furans, and, at higher temperatures, particular in supercritical water, carbon monoxide and hydrogen, syn gas, a mixture from which pretty much any commodity now provided by petroleum can be synthesized, and commodities superior to gasoline like dimethyl ether can also be synthesized.
This graphic from the first paper gives a scheme for chemicals and fuels that can be obtained from pyrolysis:

The caption:
I personally find this graphic unsatisfying - but I picked this paper for this discussion somewhat at random - because it does not discuss biomaterials, specifically the many important allotropes of carbon itself, nor does it discuss carbides. These allotropes and carbides represent the opportunity to sequester carbon at a profit.
A process schematic, one very limited in scope:

The caption:
Fixed bed reactors more or less imply batch processing, which is inherently more expensive than continuous flow systems.
Fluidized bed reactors can approach continuous processing a little closer; here's a schematic of one:

The caption:
According to the authors, the following situation applies:
I'm not sure I necessarily buy this statement - I favor syn gas above all other options - since I'm not sure it includes some consideration of very high temperatures, nor does it consider conducting reactions in supercritical oxidation systems using supercritical carbon dioxide or supercritical water.
The second paper focuses more closely on sargassum, and it is therefore more relevant to the situation now covering much of the atlantic ocean.
From the introduction:
The scientists here are Indian nationals; the previous paper was written by Iranian nationals.
The graphics in this paper address more directly what sargassum can do and the conditions for doing it:
I'll just fire these graphics off as a set:

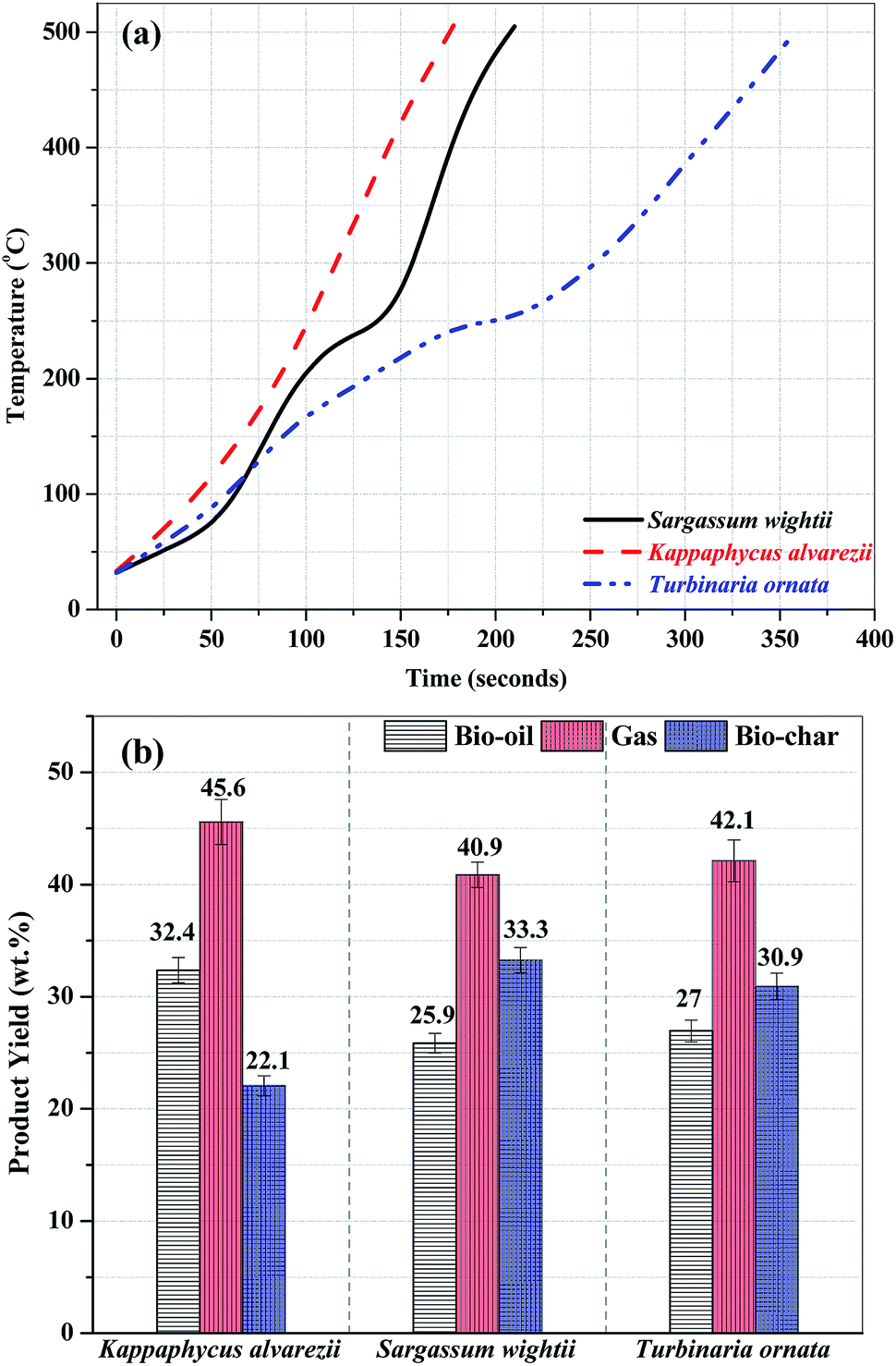
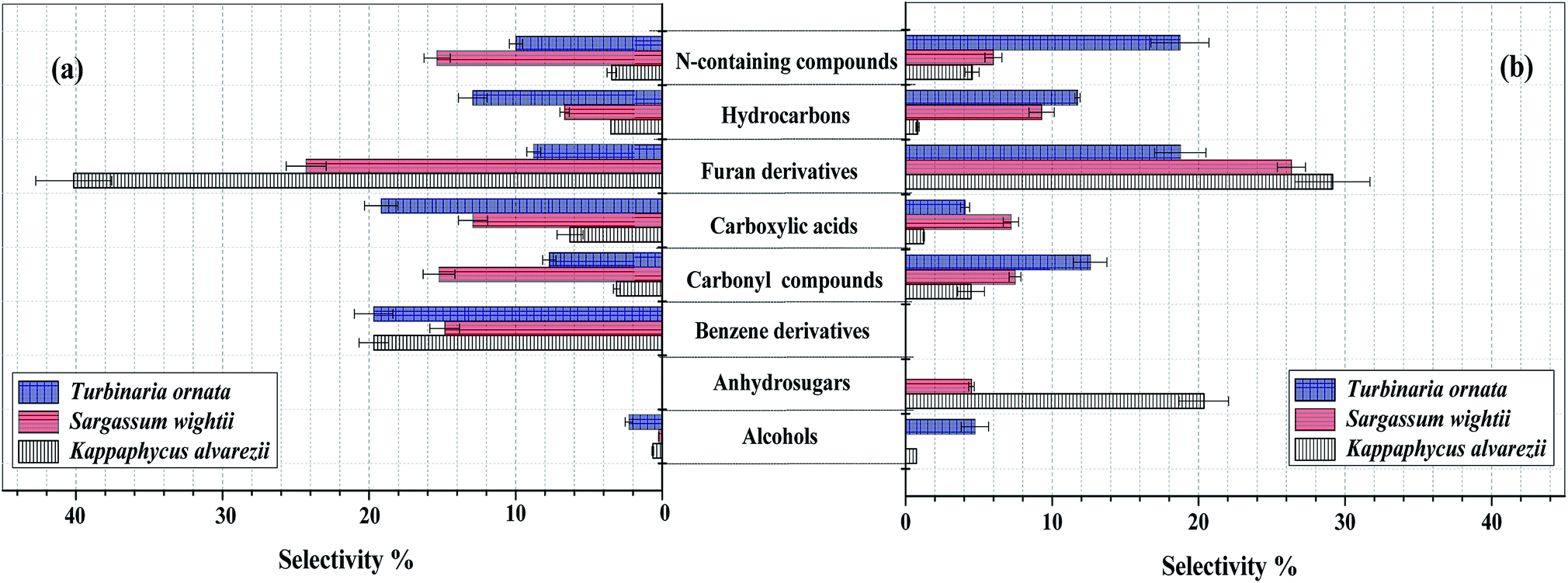
Fig. 3 (a). Composition of bio-oil from microwave-assisted pyrolysis of different macroalgae at 500 °C. (b) Composition of the pyrolysates from Py-GC/MS of different macroalgae at 500 °C. The data are presented as the mean ± S.D., n = 3.

Note that the heat source here is microwaves, and the temperature is rather low for my tastes, 500 °C. (There is brief mention of higher temperatures.)
Microwaves are featured because of their fast heating rate, but continuous heating can be achieved by gas flows, particularly with nuclear heat, which is of course not mentioned here.
Many of the chemicals shown would require further processing to make them usable as fuels, for example the furans, or as commodity chemicals. The alkanes are linear, and thus not really "gasoline-like" which is not a bad thing necessarily, since gasoline is a ridiculously dangerous and noxious fuel, one that we stupidly accept because of unwarranted (and dangerous) familiarity.
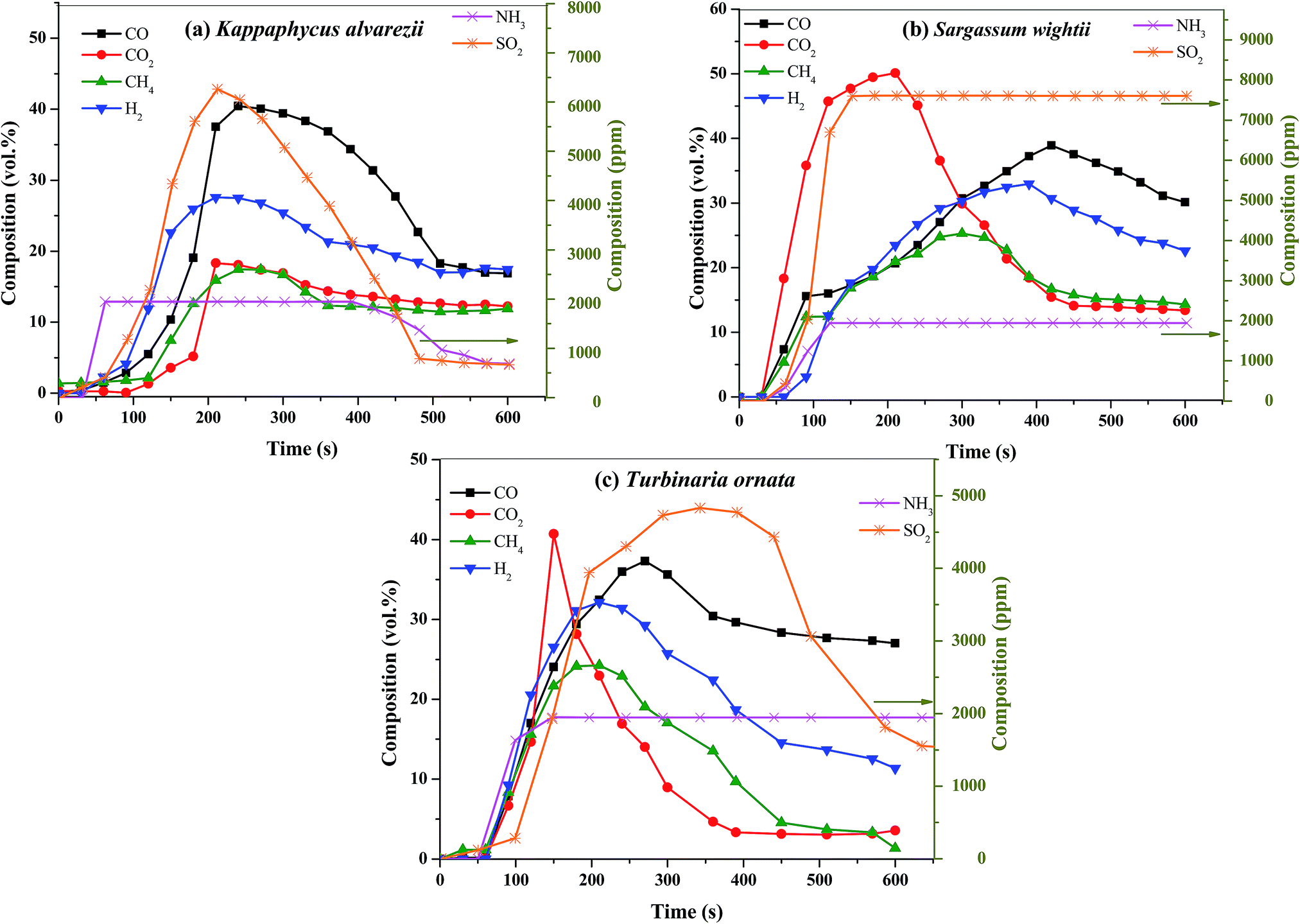
Full oxidation to syn gas is discussed only briefly, and then in the context of co-existing with bio-oils, many of which are phenolic species, somewhat corrosive and somewhat unstable oils. Here is an excerpt focusing on syn gas:
Sargassum contains less sulfur than the other two species investigated here. It also produced higher amounts of furans, which are potentially direct use biofuels, but more importantly, can be utilized for the production of certain kinds of interesting polymers which, in long term uses (as opposed to the noxious practice in which most of us are more or less forced to participate, single use plastics) represent carbon sequestered from the air, indirectly via seawater.
(Syngas, via the Boudouard reaction after hydrogen removal, can be made into carbides.)
I know I'm rambling here, and I'm tired, and will cut it here, but this type of process is interesting, given the sargassum catastrophe in the Atlantic ocean which will get worse before it gets better. It is only really useful if the sargassum is mined using nuclear powered ships - we know well how to make these - and pyrolyzed using nuclear reactors.
Without this, it's probably just another useless treadmill, like the rest of the so called "renewable energy" schemes.
Enjoy the rest of the workweek.
cstanleytech
(26,273 posts)as well as the increasing acid levels? Assuming both are not problems for it then it has promise I suppose but personally I think the biggest threat is not the increased carbon levels (assuming they level off of course) nor is it power generation rather the biggest threat to the world is the continuously increasing worldwide population level.
NNadir
(33,509 posts)...destruction.
It is the product of the release of nutrients from the Amazon basin now being destroyed and carbon dioxide.
I agree that the world population is unsustainable, but I note that birth rates are higher in poor countries than in rich countries, one reason being high infant mortality.
I considered this some time back on another website: Current Energy Demand; Ethical Energy Demand; Depleted Uranium and the Centuries to Come
Eko
(7,272 posts)"from the kinds of asses who write stuff - this actually happened - about the collapse of a tunnel at the Hanford weapons plant being a tragedy, while the heat deaths across Europe is regrettable but acceptable."
Certainly not here. https://www.democraticunderground.com/1127109489
If anyone thinks NNadir is speaking truth check out that thread. Its very informative.