Welcome to DU!
The truly grassroots left-of-center political community where regular people, not algorithms, drive the discussions and set the standards.
Join the community:
Create a free account
Support DU (and get rid of ads!):
Become a Star Member
Latest Breaking News
General Discussion
The DU Lounge
All Forums
Issue Forums
Culture Forums
Alliance Forums
Region Forums
Support Forums
Help & Search
General Discussion
Related: Editorials & Other Articles, Issue Forums, Alliance Forums, Region ForumsCoronavirus Test Kits and How The Test Is Done
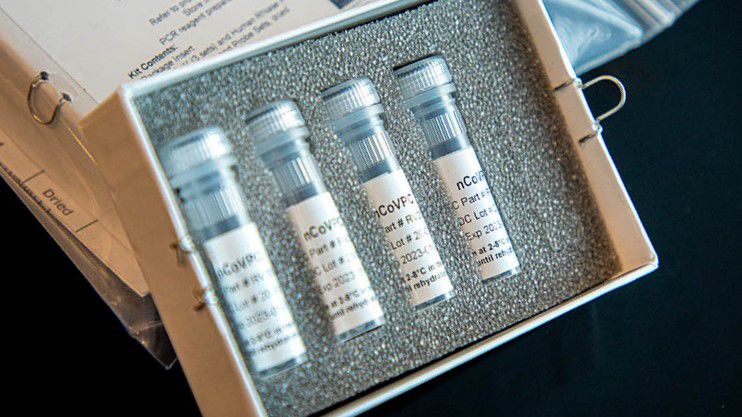
Hoping this is a photo of the correct COVID-19 test and not the ones the CDC labeled inadequate.
As of late Friday morning, The CDC website indicated that 451 coronavirus tests had been conducted nationwide.
What is a coronavirus test like? How are CDC kits handled? Here are the details.
WHAT DOES A CORONAVIRUS TEST KIT LOOKS LIKE?
The CDC on its website provides a photo of the lab test kit for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) the disease named by the International Committee on Taxonomy of Viruses that is caused by the novel coronavirus, also known as COVID-19.
The kit itself contains four small vials inside of a white box of packaging, all of which is bagged in plastic.
WHAT DOES A CORONAVIRUS TEST INCLUDE?
There are two main elements to a COVID-19 test, according to interim guidelines shared publicly by the CDC to clinicians.
The coronavirus is a respiratory disease, so the test kits take specimens from both the upper and lower respiratory tracts.
-- Tests in the lower respiratory tract include a branchoalveolar lavage, a minimally invasive procedure in which the clinician places a bronchoscope through the patient’s mouth or nose and into the lungs. A sterile fluid solution is squirted into a small part of the lung, and a specimen is collected for examination.
The patient then produces a sputum sample: “Have the patient rinse the mouth with water and then expectorate deep cough sputum directly into a sterile, leak-proof, screw-cap sputum collection cup or sterile dry container,” the CDC’s instructions read.
-- In the second test, for the upper respiratory tract, the clinician will swab the patient’s nostril as well as the throat. The test also collects a 2 to 3 milliliter specimen following a nasal “wash” or aspirate, which involves a soft, flexible tube in the nose.
WHAT HAPPENS TO THE TEST KIT AFTER THAT?
These specimens are refrigerated until they can be shipped to the CDC for testing.
The kit is to be stored at 35-46 degrees Fahrenheit and kept at that temperature using an ice pack or dry ice as it is shipped to a CDC lab.
HOW DO DOCTORS DECIDE GETS TESTED?
As of this week, healthcare providers have been testing patients for coronavirus only if they met the CDC’s set criteria for that patient to be considered a “person under investigation,” or PUI.
There are now three criteria that can be met to warrant lab testing, according to the CDC website. The third item was added Thursday under revised guidelines, as CDC Director Robert Redfield explained to congressional leaders.
-- Symptoms that include fever or coughing and shortness of breath, along with the epidemiological risk of having close contact with a lab-confirmed COVID-19 patient;
-- Symptoms that include fever and a cough or shortness of breath, along with a history of recent travel to coronavirus hot zones; or
-- A fever and severe symptoms of lower respiratory illness, such as pneumonia, that require hospitalization, even with no source of exposure via travel or contact with a confirmed COVID-19 patient.
HOW LONG DO THE TEST RESULTS TAKE?
The CDC does not specify the time frame for which results should be expected, but its guidelines call for the samples to be shipped overnight.
In the case of the patient being treated at UC Davis Medical Center, the positive diagnosis appears to have been received about three days after testing.
The CDC on its website provides a photo of the lab test kit for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) the disease named by the International Committee on Taxonomy of Viruses that is caused by the novel coronavirus, also known as COVID-19.
The kit itself contains four small vials inside of a white box of packaging, all of which is bagged in plastic.
WHAT DOES A CORONAVIRUS TEST INCLUDE?
There are two main elements to a COVID-19 test, according to interim guidelines shared publicly by the CDC to clinicians.
The coronavirus is a respiratory disease, so the test kits take specimens from both the upper and lower respiratory tracts.
-- Tests in the lower respiratory tract include a branchoalveolar lavage, a minimally invasive procedure in which the clinician places a bronchoscope through the patient’s mouth or nose and into the lungs. A sterile fluid solution is squirted into a small part of the lung, and a specimen is collected for examination.
The patient then produces a sputum sample: “Have the patient rinse the mouth with water and then expectorate deep cough sputum directly into a sterile, leak-proof, screw-cap sputum collection cup or sterile dry container,” the CDC’s instructions read.
-- In the second test, for the upper respiratory tract, the clinician will swab the patient’s nostril as well as the throat. The test also collects a 2 to 3 milliliter specimen following a nasal “wash” or aspirate, which involves a soft, flexible tube in the nose.
WHAT HAPPENS TO THE TEST KIT AFTER THAT?
These specimens are refrigerated until they can be shipped to the CDC for testing.
The kit is to be stored at 35-46 degrees Fahrenheit and kept at that temperature using an ice pack or dry ice as it is shipped to a CDC lab.
WHERE DO THE KITS GET TESTED?
The CDC on its website says laboratory test kits are shipped “to laboratories CDC has designated as qualified, including U.S. state and local public health laboratories, Department of Defense (DOD) laboratories and select international laboratories,” the latter of which include facilities with the World Health Organization and Global Influenza Surveillance Response System.
The agency said this week that a protocol developed by the CDC and the U.S. Food and Drug Administration, which “uses two of the three components of the original CDC test kit” to detect for the virus, will allow 40 additional public health labs to begin testing.
CA Governor Gavin Newsom said in Thursday’s news conference that the California Department of Public Health would be among those sites performing tests.
The CDC on its website says laboratory test kits are shipped “to laboratories CDC has designated as qualified, including U.S. state and local public health laboratories, Department of Defense (DOD) laboratories and select international laboratories,” the latter of which include facilities with the World Health Organization and Global Influenza Surveillance Response System.
The agency said this week that a protocol developed by the CDC and the U.S. Food and Drug Administration, which “uses two of the three components of the original CDC test kit” to detect for the virus, will allow 40 additional public health labs to begin testing.
CA Governor Gavin Newsom said in Thursday’s news conference that the California Department of Public Health would be among those sites performing tests.
HOW DO DOCTORS DECIDE GETS TESTED?
As of this week, healthcare providers have been testing patients for coronavirus only if they met the CDC’s set criteria for that patient to be considered a “person under investigation,” or PUI.
There are now three criteria that can be met to warrant lab testing, according to the CDC website. The third item was added Thursday under revised guidelines, as CDC Director Robert Redfield explained to congressional leaders.
-- Symptoms that include fever or coughing and shortness of breath, along with the epidemiological risk of having close contact with a lab-confirmed COVID-19 patient;
-- Symptoms that include fever and a cough or shortness of breath, along with a history of recent travel to coronavirus hot zones; or
-- A fever and severe symptoms of lower respiratory illness, such as pneumonia, that require hospitalization, even with no source of exposure via travel or contact with a confirmed COVID-19 patient.
HOW LONG DO THE TEST RESULTS TAKE?
The CDC does not specify the time frame for which results should be expected, but its guidelines call for the samples to be shipped overnight.
In the case of the patient being treated at UC Davis Medical Center, the positive diagnosis appears to have been received about three days after testing.
https://www.sacbee.com/news/local/health-and-medicine/article240733946.html?fbclid=IwAR2PrJ4DMvuWQiuHh-9xDqbCFONm5zPUPh-jQMLeMLY2v9FCgEe1e8aBxNo#storylink=cpy
InfoView thread info, including edit history
TrashPut this thread in your Trash Can (My DU » Trash Can)
BookmarkAdd this thread to your Bookmarks (My DU » Bookmarks)
4 replies, 1170 views
ShareGet links to this post and/or share on social media
AlertAlert this post for a rule violation
PowersThere are no powers you can use on this post
EditCannot edit other people's posts
ReplyReply to this post
EditCannot edit other people's posts
Rec (2)
ReplyReply to this post
4 replies
 = new reply since forum marked as read
Highlight:
NoneDon't highlight anything
5 newestHighlight 5 most recent replies
= new reply since forum marked as read
Highlight:
NoneDon't highlight anything
5 newestHighlight 5 most recent replies
Coronavirus Test Kits and How The Test Is Done (Original Post)
ancianita
Feb 2020
OP
BigmanPigman
(51,591 posts)1. CNN just reported that CDC said if you get tested by mid-morning
they will have the results by the end of the same day.
ancianita
(36,055 posts)2. Great, multiple labs will make this go faster per CDC's protocol. The #1 problem: test kit shortage.
TexasBushwhacker
(20,190 posts)3. Wow, pretty invasive n/t
Response to TexasBushwhacker (Reply #3)
ancianita This message was self-deleted by its author.