Science
Related: About this forumA Nice Scientific Review Article on the Destruction of Persistant Perfluoroorganic Pollutants.
The paper I'll discuss in this post is this one: Destruction of Per- and Polyfluoroalkyl Substances (PFAS) with Advanced Reduction Processes (ARPs): A Critical Review (Junkui Cui, Panpan Gao, and Yang Deng, Environmental Science & Technology 2020 54 (7), 3752-3766). Although EST is primarily a journal that reports on original research, it also features some viewpoint, policy and, as in this case, review articles.
Review articles, as every scientist knows, refer to other papers in the scientific literature to generate the authors' consensus on the state of a particular area of research.
The situation with respect to the widespread, and highly intractable PFAS (perfluoroalkyl substances) contamination in the environment is a very hot topic because these synthetic molecules were and are widely used. The most familiar perfluoroalkyl substance that most people have in their homes, often in multiple settings, is Teflon. Teflon when overheated can degrade into volatile perflouro compounds, which are known to kill, for example, caged birds in people's homes. It is generally not fatal to human beings of course, but being non-fatal does not imply that ingestion of perfluoro compounds is good for you. There's a lot of evidence in fact that it's not good for you. Perfluorocompounds have also been widely used in many other types of commercial products, spray on fabric protectors for furniture (although these products, including Scotch Guard, are no longer available), fire fighting foams, where massive amounts were sprayed on fires to put them out, electronic devices, and hydrogen fuel cells. As a result, they are widely distributed in water, ground, and biological flesh, including human biological flesh, where they are thought to represent a growing risk. I will briefly refer to the text of the review article & a few interesting graphics here and then comment on a few papers I found referenced in the review, as well as a few I came across in references in the references.
I am very interested in this topic because I am very interested in the utilization of radiation resources, because it turns out that radiation is a very potent tool for addressing this otherwise intractable problem.
The introductory text of the review gives a nice overview of the problem:
However, concerns on PFAS have gradually been growing due to their prevalence, mobility, persistence, bioaccumulation, and adverse health effects.(2,5?9) Many PFAS have been demonstrated to bioaccumulate, can bind to blood proteins, and have long half-lives in humans. Human PFAS exposure is linked to cancer, obesity, elevated cholesterol, immune suppression, and endocrine disruption.(10?12) Various PFAS, particularly perfluorooctanoic acid (PFOA) and perfluorooctanesulfonate (PFOS), have been frequently identified in surface freshwater,(13?15) groundwater,(7,16?19) drinking water,(20?22) and landfill leachate.(23?27) The emerging anthropogenic chemicals are challenging water and wastewater treatment, water reclamation, site remediation, and landfill leachate disposal because of a lack of effective, efficient, and practical treatment technologies. Meanwhile, a tremendous pressure potentially derives from the regulatory determination. In 2016, US Environmental Protection Agency (EPA) issued a Lifetime Health Advisory (LHA) for PFOA and PFOS at 70 ng/L.(21) Presently, US EPA is moving forward with the maximum contaminant level (MCL) process for PFOA, PFOS, and probably more PFAS chemicals in drinking water.(28) The pursuit of public health and potential regulatory pressure require the water industry to stay current and proactive for advancing innovative PFAS treatment technologies. Among very few technically effective PFAS treatment methods, advanced reduction processes (ARPs) have recently emerged as a promising option.
Note that the remark about the EPA is historic, and refers to a historical time when there was actually a President of the United States who did not hate his country and its citizens, who actually cared about their health and in fact whether they lived or died.
Obviously this situation is no longer obtained. The country is being run by a group of very nasty and ignorant people known as Republicans, who couldn't care less about whether Americans are healthy, safe or live or die, so long as their donors and themselves can make money and worship an intellectually and morally paralytic as part of a twisted and sick religion. Apparently in their "faith" fetuses, and of course, guns, are more important than living breathing people.
This however, is the science forum, not the religion forum, so excuse the aside.
A covalent chemical bond that exhibits cylindrical symmetry around the bond axis and exhibits high electron density in the region between the atoms in the bond is known as a "? bond." Among ? bonds, because of the high electronegativity of fluorine - the highest in the periodic table - the carbon fluorine bond is the third strongest, after the boron fluorine bond and the silicon fluorine bond. (The nitrogen fluorine bond is also very strong, NF3, a greenhouse gas, is now found in the atmosphere and its concentration, like many other fluorine containing gases including hydrofluorocarbons, sulfur hexafluoride and related compounds, is rising.)
To break a carbon fluoride bond, therefore requires significant energy.
If we take the bond dissociation energy of the carbon fluorine bond from this table, 536 kJ/mole, and then calculate using Avogadro's number, the unit electric charge, and Planck's constant the frequency of light that has the energy to dissociate this bond, we find that the frequency is around 1.3 PHz. (PetaHertz), or 1.3 X 10^(15) Hz, corresponding to a wavelength of around 220 nm. This is in the ultraviolet region, specifically the UVC region, which is the wavelength of UV radiation is used to sterilize objects, is carcinogenic since it also breaks bonds in skin, and is filtered out largely by the ozone layer. Without such filtering terrestrial life could not exist.
Period.
However, simply breaking this bond with light at this wavelength does not necessarily prevent the bond from reforming, and thus it is not the case that simply irradiating PFAS, even with UV radiation, destroys them. This is also observed in the case of the breaking of chlorine or bromine carbon bonds in the upper atmosphere, and accounts for the fact that CFC's have persisted a long time, being effectively catalytic owing to recombination of the original chlorine (or bromine) radicals after they go through happily destroying oodles and oodles of ozone molecules in chain reactions. Relatively rare reactions do terminate these chain reactions in such a way as to lead to the destruction of CFCs them over time. These reactions are rare in the upper atmosphere, because water is very dilute there: It is the reaction with radicals derived from water that cause the decomposition of CFC's, with the chlorine or bromine ending us as either hydrochloric or hydrobromic acids. Happily these destructive CFC or Brominated carbons are faster than is the case for HFC's, which - unless something is done to change the situation - they will persist for tens of thousands, even hundreds of thousands of years. The lifetime of most CFC's is thought to be decades or hundreds of years.
PFAS in water and soil are not exposed to UV radiation except in industrial settings, thus they will persist for a very, very, very long time.
There have been a huge number of papers in the scientific literature on the subject of PFAS, which is a good thing, since it is a very serious matter, many on destroying them by various chemical means, and but the paper here focuses on a particular approach - it may be the best approach, which is reductive processes, what is called in the paper "advanced" reductive processes. A reducing agent is anything that gives up an electron readily. Alkali metals, which are very reactive, are all reducing agents, cesium metal being the strongest among them. When dissolved in liquid ammonia, pure ammonia and not the familiar household aqueous solution, these metals actually produce one of the most powerful reducing agents, the solvated electron.
It is also possible to create solvated electrons in water, using - this is unsurprising - UV (or higher energy) radiation which causes water to dissociate into hydrogen radicals and hydroxide radicals, the latter being one of the most powerful oxidizing agents.
This graphic from the article is a schematic cartoon showing the way that solvated electrons interact with PFAS to decompose them in multiple steps. The graphic will also show, besides free electron, the present of sulfur and iodine species which, as discussed in the review article, are actually designed to generate free electrons. To wit, from the text:
The caret ( ^ ) here is used to designate superscripts which are no longer available in the DU editor.
The iodine and sulfur species can react with the generated (destructive) PFAS radicals to form iodinated or sulfinated species which are far less persistent, and far easier to destroy than PFAS themselves, often by biological or readily available chemical means, and thus do not represent quite the same risk as the PFAS.
This is shown in the graphic from the paper:
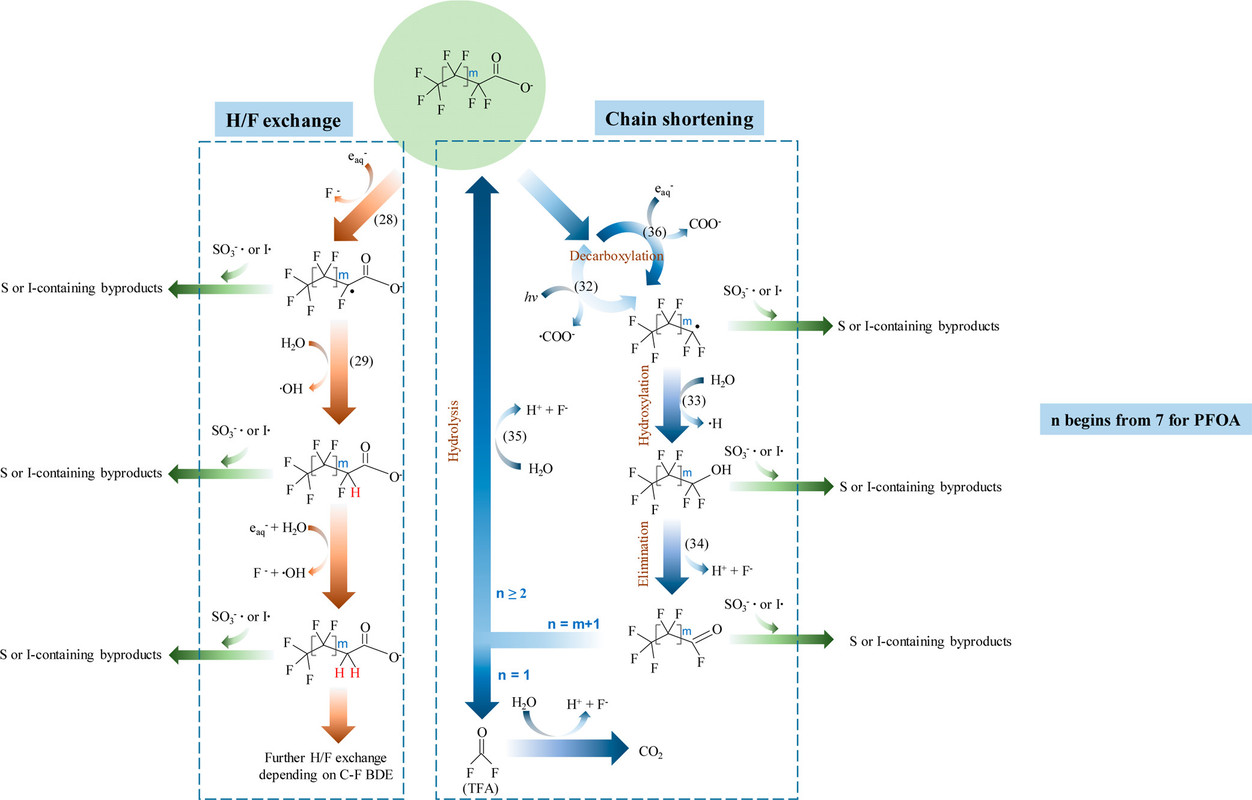
The caption:
PFOA is a very common PFAS, found in many products. PFOA is the abbreviation of perfluorooctanoic acid. The destruction of PFOA can proceed by both pathways, the one on the left and the one on the right.
Note that there is an error in this diagram. The molecule on the very bottom of the right identified as "TFA" which generally stands for the common reagent trifluoroacetic acid, is not TFA. It is fluorophosgene, a very toxic molecule which thankfully has a very short half-life in water, especially in basic solution. (The common fun reagent known as "phosgene" - used in the first world war as a war gas - is chlorophosgene.)
Fluorophosgene can also be produced from the persistent greenhouse gas tetrafluoromethane, which is a side product of the aluminum industry when carbon electrodes made from petroleum coke are oxidized by electrochemically generated fluorine gas in the Hall process. In considering the radiochemical destruction of this gas, I was surprised to learn a few years back that a precursor of fluorophosgene, trifluoromethanol is surprisingly long lived; I'd assumed that its decomposition to fluorophosgene was very fast, but apparently I was wrong. I believe that trifluoromethanol can be isolated although it would definitely be unpleasant stuff to be around, since ultimately it will decompose to fluorophosgene.
Note that all of these pathways lead to the generation of HF, highly corrosive hydrofluoric acid. However, in these pathways, the concentration of this acid (or its gaseous form) happily remains relatively low. Both HF and fluorophosgene are conveniently destroyed by bases; calcium hydroxide being one of the best, since it produces insoluble calcium fluoride. I performed this neutralization many times myself when I was a kid; it works great.
Another graphic shows a similar set of pathways for the decomposition for another major component of PFOS, which is also abbreviated "PFOS" but where the "OS" means "octanoyl sulfonate" rather than "organic substances." Perfluorooctanoyl sulfonate is another widely distributed product found, like PFOA, in ground water, river water, and living things.
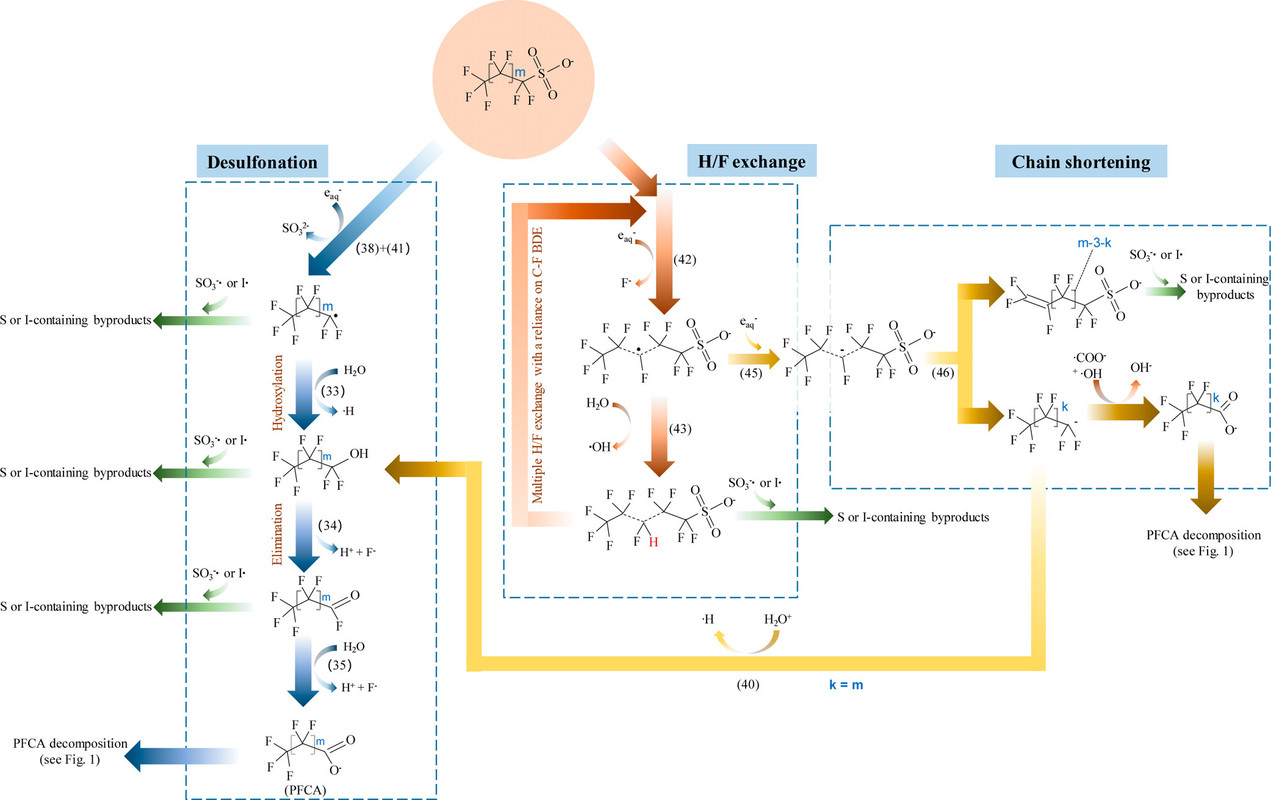
The caption:
In this case they have avoided the possible confusion about the meaning of the PFOS abbreviation by referring calling them perfluorosulfonic acids.
Reference 68 in this paper is this one:
The Hydrated Electron (Herbert and Koons, Annual Review of Physical Chemistry Vol. 68:447-472 (2017)).
I referenced in another post in this space, a quick sidelight referring because of an interesting historical note in it, but the thread turned annoying in such a way that my sense of humor deteriorated, something easy to do.
Nevertheless, the paper has an interesting graphic that shows the pathway of generation of solvated electrons:
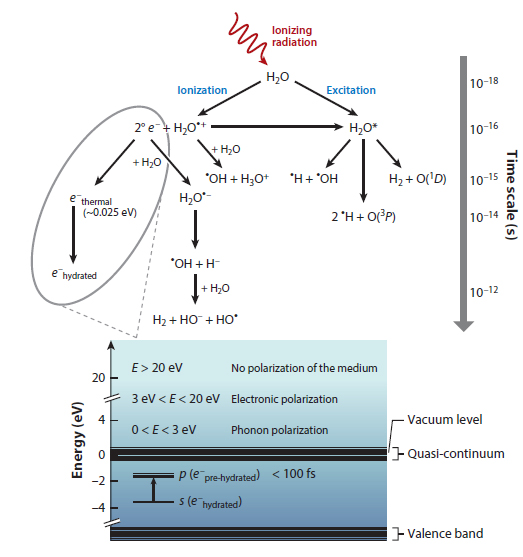
The caption:
In the context here, the radiation is UV radiation, although it need not be, higher energy radiation will work, a subject to which I'll return below.
Reference 47 in this paper just cited is, by the way, this one: Precursors of Solvated Electrons in Radiobiological Physics and Chemistry (Elahe Alizadeh and Léon Sanche, Chem. Rev. 2012, 112, 5578?5602)
When I went to open it, I noticed that the particular issue of Chemical Reviews, which is about 8 years old, had a number of review papers on solvated electrons, and I poked around in a few of them. A paper not specifically about solvated electrons per se, but certainly touching on them concerned the degradation of Nalfion membranes in hydrogen fuel cells, which are often represented as being "green" although Nalfion is a perfluorinated polymer and there is nothing, despite much thermodynamically illiterate nonsense one can read everywhere, including in the scientific literature, about hydrogen that is green. Almost all of the hydrogen produced on an industrial scale is made by utilization of the dangerous fossil fuel dangerous natural gas.
Nevertheless, these fuel cells are commercial, but are utilized mostly as back up power for critical systems in power outages, cell phone towers, medical facilities and the like.)
The paper on their degradation is this one:
Fuel Cell Perfluorinated Sulfonic Acid Membrane Degradation Correlating Accelerated Stress Testing and Lifetime (Marianne P. Rodgers,* Leonard J. Bonville, H. Russell Kunz, Darlene K. Slattery, and James M. Fenton, Chem. Rev. 2012, 112, 6075?6103). The discussion in the paper refers to the decomposition products of Nalfion, one of which is hexaflouroacetone. Another is hydrofluoric acid. Hexafluoroacetone is often utilized in analytical chemistry in the HPLC and LC/MS/MS of nucleic acids and therapeutics derived from nucleic acids. When it is, it requires special precautions on the part of the laboratory science since hexafluoracetone is an extremely toxic compound, if I recall correctly is a powerful hepatotoxin. I am aware of many labs that have refused to work with the compound, although obviously some do.
Nevertheless, hydrogen fuel cells are "green." So I've heard.
By the way, people who are always prattling on about "green cars," an oxymoron if there ever was one since there is no way to make the car CULTure sustainable, often talk about hydrogen fuel cell cars, and in fact, this is a topic of the Chemical Review paper just linked, how to make fuel cells for cars.
Returning to the subject of the more common perfluorocompounds addressed in the opening post, a point made in some of the references and in other papers I've read independently on this topic is that the solvated electron, a reducing agent, is very much the main species responsible for the degradation of PFOAs. Interestingly solvated electrons are produced in the same pathway as the powerful oxidizing agent, the hydroxide radical, as mentioned above.
Obviously the recombination of these species can lead to lower effectiveness for radiation's effects on PFOA. There is a way to shift the equilibrium toward solvated electrons as opposed to OH and H radicals, and this is via the use pH.
This is discussed in this paper: Effect of initial solution pH on photo-induced reductive decomposition of perfluorooctanoic acid (Yan Qu, Chao-Jie Zhang ?, Pei Chen, Qi Zhou, Wei-Xian Zhang, Chemosphere 107 (2014) 218–223. The authors find a profound effect of pH, with the hydrated electron surviving longer at high pH. The UV wavelength they were utilizing was 254 nm, somewhat longer than the 223 nm wavelength I calculated above, although this wavelength was generalized to a "typical" C-F bond and not necessarily the wavelength in say, PFOA. The solvated electrons are generated via an iodide intermediate.
The authors propose the following rationale for the effect of pH that they find:
Eqn 8:

Electronic repulsion between the negatively charged hydroxide and the charged solvated electron and differences in the hydration spheres of the various species prevent similar addition to the OH- ion to quench electrons by formation of the hydroxide radical which in any case, were it to exist, be doubly charged.
It should be said that in this particular setting, the authors noted the presence of perfluorinated gases such as CF4, C2F6, etc, so there's that. It also must be said that they needed to rigorously exclude oxygen from the system.
The importance of hydration spheres on kinetics involving solvated electrons is discussed in this paper: Hydrogen Forms in Water by Proton Transfer to a Distorted Electron (Jungwirth, et al., J. Phys. Chem. B 2010, 114, 2, 915-920)
Japanese authors have noted that pH has little effect in the presence of organic molecules in the system: Factors influencing UV photodecomposition of perfluorooctanoic acid in water (Giri et al., Chemical Engineering Journal 180 (2012) 197– 203) An interesting point made in this paper is the influence of wavelength. Standard UV lamps (Hg lamps) radiate at the 254 nm mid UV range. Diode array instruments, widely used in analytical chemistry can reach down to around 200 nm, but the intensity of these beams is not required to be very high, and there is a point at which glass absorbs UV. The authors in this paper used a special kind of synthetic glass that was designed to transmit the higher energy shorter wavelength 185 nm light.
This led to a dramatic change in the performance of the system in degradation as shown:

The caption:
Of course, there is radiation that is much more energetic than 185 nm wavelength energy, specifically X-rays and gamma rays. These radiations can produce very high energy electrons either through photoelectric or the related Auger process besides directly scissioning carbon fluorine bonds.
A review article going back some 15 years ago on the subject of electrons in water, explicitly covers very high energy electrons, whereas the other papers discussed in this thread discusses moderately high energy electrons, those in the 5-6 eV range, the UV range. In all cases, the papers discuss the thermalization of electrons at these energy, thermalization being the process by which their energy is reduced to levels associated with the kinetic energy of molecules to those associated with ambient temperatures.
This paper is here: Role of Water in Electron-Initiated Processes and Radical Chemistry: Issues and Scientific Advances (Chem. Rev. 2005, 105, 355-389) It's written by a consortium of scientists representing a fair percentage of the major radiation laboratories in the United States, both National Labs and Academic Institutions.
The point here is that highly energetic electrons vastly enrich the number of electrons, and thus the rate of decomposition of analytes reacting with the rich array of reactions initiated by these electrons.
Figure 1 from this paper shows that it all begins when ionizing radiation produces electrons, the more energetic the electrons will be.
Figure 1:
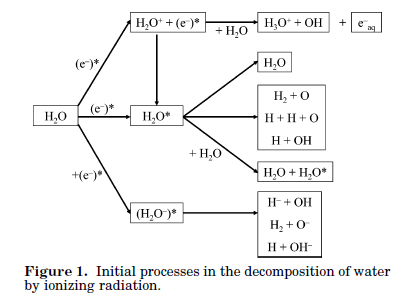
The caption:
The richness of the chemistry of water in a radiation field is given by a table from this paper:

Of course many of the species here are highly reactive in their own right, and many would serve to degrade many contaminants in dangerously degraded water, including, but not limited to PFOSs.
Cesium hydroxide is the strongest base known in aqueous solution and I have gone so far as to contemplate a solution of highly radioactive cesium hydroxide in which the radioactive cesium was added directly, as the hydroxide, to severely contaminated water. In the PFAS case, there are many examples of groundwater of this type, particularly in the upper midwest. However, the amount of the most readily available highly radioactive isotope of cesium, Cs-137, is relatively small. A crude calculation on my part, based on a simplified solution to the Bateman equation, the simplification justified by the low neutron capture cross section of Cs-137 and the fact that nuclear power has been producing about 28 exajoules of energy per year suggests that on the entire planet there is only 200 - 300 metric tons of this isotope, a tiny amount when compared to the sheer volume of highly contaminated water on this planet.
Moreover, the cesium-137 available in used nuclear fuel is not isotopically pure, it is contaminated by the non-radioactive isotope Cs-133 (cesium's naturally occurring isotope which is also a fission product), very small amounts of cesium-134, a short lived isotope formed (half-life 2.06 years) by neutron capture in Cs-133, cesium-135, a very long lived radioactive isotope whose long half-life limits its use as a radiation source, and cesium-137.
A French paper some years back which utilized some elegant approaches to separate cesium isotopes from their non-radioactive barium daughters, gave some insight to the relative proportions of these isotopes in used nuclear fuel, by dissolving a used nuclear fuel pellet in nitric acid and using an analytical technique known as ICP/MS to measure the proportions of cesium isotopes in them, barium free, a neat trick. That paper is here: Cs–Ba separation using N2O as a reactant gas in a Multiple Collector-Inductively Coupled Plasma Mass Spectrometer collision-reaction cell: Application to the measurements of Cs isotopes in spent nuclear fuel samples (Granet et al., Spectrochimica Acta Part B 63 (2008) 1309–1314).
Here is a mass spectrum showing the cesium isotope distribution in freshly isolated nuclear fuel:

The caption:
There is no information about the cooling period this fuel pellet underwent, but it reasonable to assume that it wasn't all that long. Consider a used nuclear fuel that we might have in New Jersey, fuel removed from the Oyster Creek Nuclear Reactor in 1971, two years after the reactor came on line. (It closed last year, regrettably.) This fuel is 59 years old. The half-life of cesium-137 is 30.08 years. This means that only 25.6% of the original cesium-137 is still radioactive and that if the fuel's distribution looked similar to that in the French graph, the Cs-137 peak is only 1/4 the size it was back then. This means that the radioactivity is diluted significantly.
In recent years, I've come back to an idea that I abandoned when I knew far less about nuclear engineering than I know now, which is to put cesium salts into certain chambers in a nuclear reactor to conduct certain kinds of heat transfer tasks coupled with neutronics control tasks. The non-radioactive isotope of cesium, Cs-133, has more and stronger neutron capture cross sections in the epithermal region than any of the radioactive isotopes, meaning that it is readily possible to increase the radioactivity of "old" decayed cesium to make it highly radioactive because of the short half-life (and high energy) of the Cs-134 isotope to assist residual Cs-137 that has decayed even as much as 1971 Oyster Creek fuel.
Here is the neutron capture spectrum over the fission neutron range:

Even in the happy case where we produced all of our energy by nuclear means, we would only be producing less than 300 tons of fuel per year, the new accumulations being reduced each subsequent year as we approach ever closer to the Bateman equilibrium limit.
Nevertheless, the seriousness of the PFAS contamination levels on this planet which have hardly arrested, suggests we ought to consider something along these lines, assuming that we can get dumb guys and gals to reject their very dangerous fear and ignorance about radiation.
This is a long post, but I had some fun writing it, so I hope no one minds.
I wish you a safe and healthy week, and some pleasure from "making the best of it."
eppur_se_muova
(36,262 posts)It's been known for a long time that acid fluorides react with an F- catalyst to form perfluoroalkoxides which react with F2 in situ to form hypofluorites. This paper reports on salts formed from non-coordinating cations. https://pubs.acs.org/doi/abs/10.1021/acs.orglett.0c00099
(The Wikipedia article on HOF and deriviatives is very brief. I was gratified to spot the familiar name of Shlomo Rozen, one of whose lectures on such species I enjoyed attending.)
NNadir
(33,518 posts)I've collected a number of papers on the subject, once I learned of it.
It does, however, decompose to fluorophosgene and HF however, but at appropriate temperatures and pressures under anhydrous conditions it is metastable and can be isolated.
Fluoroform is, for reasons I'm not sure I understand, not quite as acidic as chloroform. This of course is problematic.
A side product of aluminum manufacture is the disturbing tetrafluoromethane. Radiation is a sink.